 
This results in a lower density of warm water. In contrast, warm water has more energy, causing water molecules to move more quickly and take up more space. This means that they can pack more closely together, resulting in a higher density. Why is cold water denser than warm water?Ĭold water is denser than warm water because as water molecules cool down, they lose energy and slow down. 
As the temperature of water increases or decreases from 4 degrees Celsius, its density will change accordingly. At 4 degrees Celsius, water is at its densest, with a density of 1000 kg/m3. This results in an increase in the density of water. As the temperature decreases, the movement of water molecules slows down, allowing them to pack more closely together. 
Yes, water is more dense at colder temperatures. Is water more dense at colder temperatures? Another common method for measuring liquid density is a densitometer, which can measure the density of liquids over a range of temperatures using different technologies such as oscillation or resonance. Hydrometers come in various types and designs and can measure densities over a wide range of temperatures. One of the most common methods is using a hydrometer, which is an instrument that measures the density of a liquid relative to the density of water. Measuring the density of water at different temperatures is possible through a variety of methods. In the production of chemical solutions, precise measurements and dilutions require an accurate understanding of water density at various temperatures. The density of water at different temperatures affects its ability to carry heat effectively, which can impact engine performance. In the automotive industry, for example, the cooling system relies on the ability of water to carry heat away from the engine. The effect of temperature on the density of water is also critical in the fields of engineering and manufacturing. Changes in the temperature and density of water can influence ocean currents, which can, in turn, affect the global climate. 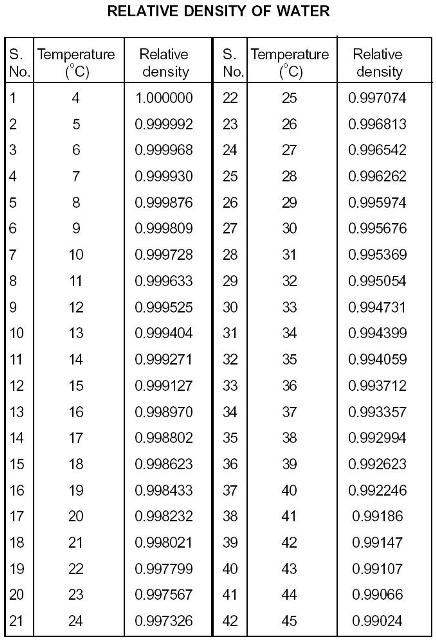
The density of water also has significant implications for the Earth’s climate and weather patterns. In hydrology, the density of water plays a vital role in the circulation and movement of water bodies such as oceans, lakes, and rivers. The impact of temperature on the density of water is an important factor in many scientific and practical applications. When water reaches its freezing point, its density increases even further, causing ice to float on top of liquid water. At 4 degrees Celsius, which is the temperature at which the density of water is the greatest, water has a density of approximately 1000 kg/m3. This results in a corresponding increase in density, which is why ice is less dense than liquid water. At 100 degrees Celsius, water has a density of approximately 958 kg/m3, which is significantly lower than the density at 0 degrees Celsius.Ĭonversely, as water cools down, the molecules in the water move more slowly, and the space between the molecules becomes smaller. This effect is particularly noticeable when water reaches its boiling point. These faster-moving molecules create more space between them, resulting in a decrease in density. As the temperature of water increases or decreases from this point, the density of water decreases.Īs water warms up, the molecules in the water become more energetic and move around more quickly. At 4 degrees Celsius, water has its maximum density of 1000 kg/m³. Temperature (☌)Īs shown in the table, water has its maximum density at 4☌, and as the temperature decreases from 4☌ to -10☌, the density of water increases by approximately 2.8 kg/m³.Īs the temperature of water increases or decreases from 4 degrees Celsius, the density of water changes accordingly. As a result, warm water will float on top of cool water. This is because the molecules in warm water are moving faster, resulting in more space between the molecules. The density of water decreases as temperature increases, meaning that warm water is less dense than cool water. How Does Temperature Affect the Density of Water? This inverse relationship between density and temperature is a fundamental principle that governs the behavior of water under different conditions. In general, as temperature increases, the density of water decreases, while the density of water increases as temperature decreases. Density refers to the amount of mass a substance has per unit of volume, and temperature has a significant effect on the density of water. Water is a unique substance that behaves in peculiar ways, particularly in terms of its density. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
